I previously worked as a peptide researcher in a university lab. Here are some things you might want to know about peptides!
What are peptides?
Peptides and proteins are made of the same building block, known as amino acids. The difference between peptides and proteins are the length of the amino acid chains. Proteins are very long chains of amino acids, usually between 200 to 600 in a chain! Peptides are much smaller, only a fragment of the size. Most peptides are between 5 to 30 amino acids in length, but they can be longer.
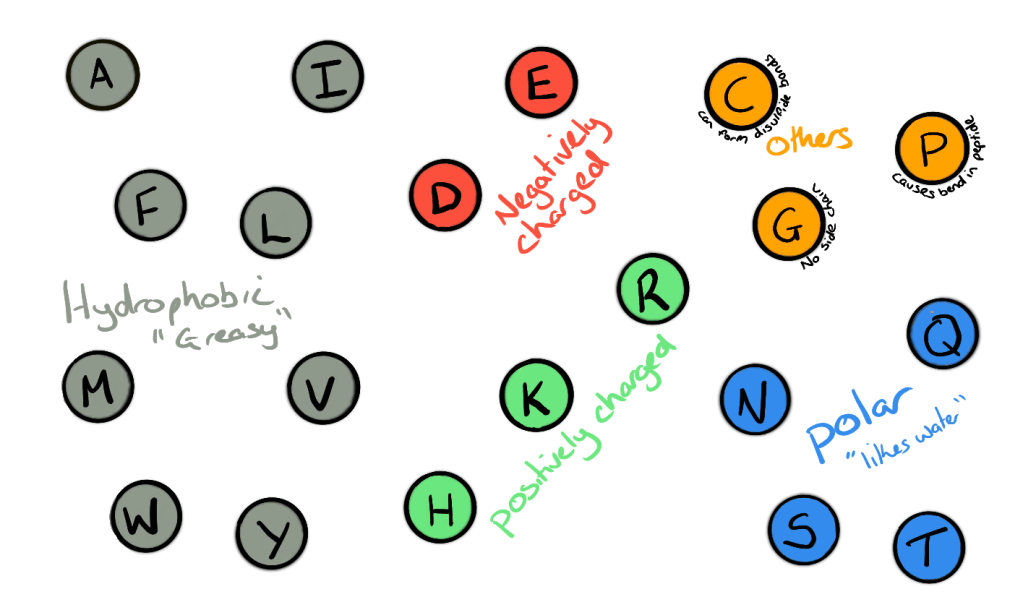
Amino acids are often represented as circles with either a single letter or three letter code inside. If we were to look at the full chemical structure of peptides and proteins it can often get confusing due to their large size. The codes let scientists know which amino acid is which without looking at the full structure. The side chains of each amino acid can be used to sort them into groups based on their properties.
How are they made?
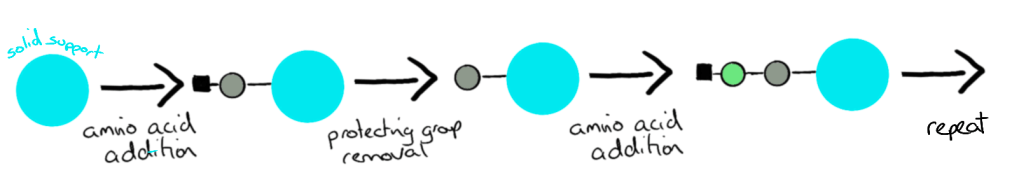
In research labs, peptides are made by a process called solid phase peptide synthesis (SPPS).
This technique allows peptides to be built on to a solid support (or resin) while washing away any of the excess chemicals used, making the process relatively simple.
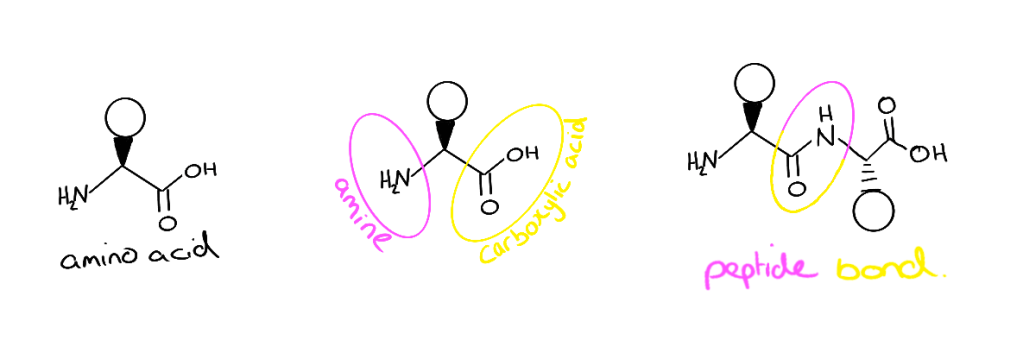
Each amino acid contains two reactive groups, a carboxylic acid and an amine. The side chains of amino acids are what makes each one different. The connection holding amino acids together in peptides and proteins is called a peptide or amide bond. It involves a carboxylic acid group of one amino acid and the amine or another. To chemically synthesise a peptide, we need to use special amino acids that have a protecting group on the amine of each amino acid. This is to stop more than one of the same amino acid attaching to the peptide.
We start by mixing the solid support, the first amino acid and coupling reagents and waiting for them to react. This cannot be seen with our eyes, we need to use special techniques to make sure the amino acid attached to the support properly. We wash away all the chemicals off the support and then it is ready for the next reaction.
Before adding the next amino acid, we need to add a chemical mixture which will remove the protection from the second reactive group (the amine). Once again, you can’t see this reaction happening, you have to make sure it is finished by special techniques.
This process repeats until you have built your peptide.
What are they used for?
The public interest in peptides has exploded in recent years as some peptide medications are becoming household names. Scientists have not always had an interest in peptides as they don’t adhere to the strict rules set for medications which can be taken as tablets.
Peptides generally break all of these rules, meaning if you take the peptide as a tablet, it will likely not be absorbed into your blood stream. There is also the added pressure that your gut is very good at breaking down peptides and proteins into amino acids, and so the peptide would be chopped up before you even have the chance to absorb it.
Scientists have been working on modifications which can be made to peptides to stop them from being chopped up. They are also often formulated as an injection rather than a tablet, to ensure it makes reaches the target area from the bloodstream.
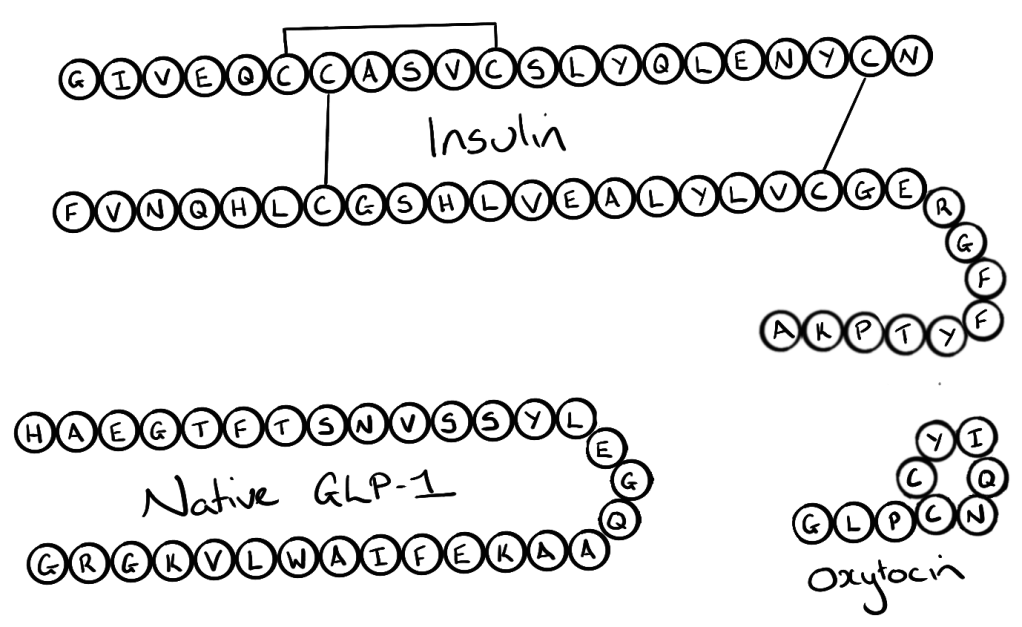
Examples of peptide medications are insulin, oxytocin and the recently described GLP-1 agonists.
Thank you for taking the time to read my blog post! Check out the posts below or head back to the blog feed by clicking here.

Leave a comment